In the present work, the saturated liquid and vapor densities of normal heptane are investigated based on a previously formulated global equation of state in the vicinity of the critical point. In particular, a crossover thermodynamic potential is used to represent accurately the liquid and the vapor experimental data of normal heptane reported by Abdulagatov and co-workers and those measured by Artyukhovskaya and coworkers as well as for information purposes for those generated from NIST REFPROP package.
The thermodynamic properties of fluid systems near the critical point has been thoroughly studied for several years [1– 3], and conducted in analogy with 3 dimensional Ising systems [4] . Universality of critical behavior of both systems results from the existence of large fluctuations in the order parameter related with the critical phase transitio n in density in one component fluids. The range of these fluctuations becomes much more significant than any microscopic scale. Therefore, the thermodynamic behavior of fluids can be characterized by scaling laws with universal critical exponents and unive rsal scaling functions of three dimensional (3D) Ising like systems [4]. Furthermore, contrary to the 3 dimensional Ising like systems coexistence curve symmetry, fluid systems demonstrate a vapor liquid asymmetry in the shape of their coexistenc e curve.
It is well established that an empirical analytic equation of state does not describe the correct thermodynamic behaviour of fluids in the critical region. In addition, an equation of state that is valid in the critical region cannot be easily ext rapolated into the classical region. Therefore, an integrated equation of state can be considered so that the thermodynamic properties of fluids over a wide range of temperatures and densities, in the critical region as well as in the classical region, far away from the critical point can be reproduced with good agreement with experimental data. An equation of state based on the crossover model formalism is already available Abbaci [5], Abbaci [6], Wilson and Kogut [7], Nicoll and Albright [8], Nicoll [9], Bagnuls and Bervillier [10], Patashinskii and Potrovskii [11].
The objective of this work is to predict the liquid and vapor densities of fluids near the critical point as well as the coexistence curve diameter of fluids. In particular, the coexistence curve diameter of n heptane will be investigated and it will be shown that we can reproduce the liquid vapor coexistence curve diameter, only by using the already formulated equation of state based on the crossover mo del for as was the case for argon Ladjama and Abbaci [12], sulfur hexafluoride Abbaci [13], ethane Abbaci, Berrezeg and Samar [14].
The modern theoretical description of systems close to the critical point is based on the renormalization group theory (RG) [8]. Specifically, different physical systems with the same space dimensionality d , a nd the same number n of components of the order parameter can be grouped within the same universality class. Based on earlier work of Nicoll J. F., and Albright [8], and Bagnuls and Bervillier [10], a crossover model based on the renormalization group theory of critical phenomena has been developed to represent the thermodynamic properties of fluids in the critical region [5, 13].
Let \(\rho\) be the density, \(T\) the temperature, \(P\) the pressure, \(\mu\) the chemical potential and A V the Helmholtz free energy per unit volume. We make these properties dimensionless with the aid of the critical parameters [5, 13]. \[\tilde{\rho}=\frac{\rho}{\rho_c},\qquad \tilde{T}=\frac{T}{T_c},\qquad \tilde{P}=\frac{P T_c}{P_c T},\qquad \tilde{\mu}=\frac{\mu T_c \rho_c}{T P_c},\qquad \tilde{A}=\frac{A T_c}{V T P_c}. \label{eq:dimless} \tag{1}\]
In addition, define \[\hat{\rho}=\tilde{\rho}-1,\qquad \Delta\tilde{T}=\tilde{T}+1,\qquad \Delta\tilde{\mu}=\tilde{\mu}_c-\tilde{\mu}_0, \label{eq:defs3} \tag{2}\] and \[\Delta\tilde{A}=\hat{\rho}\,\tilde{\mu}_0(\tilde{T})-\tilde{A}_0(\tilde{T}). \label{eq:defs4} \tag{3}\]
Here, the terms \(\tilde{\mu}_0(\tilde{T})\) and \(\tilde{A}_0(\tilde{T})\) are analytic backgrounds of functions of \(T\) subjects to the conditions that at the critical temperature, \(\Delta\tilde{\mu}(T = T_c) = 0\), \(\Delta\tilde{A}(T = T_c) = -1\). The equation of state used here is expressed in the form of a reduced Helmholtz energy which can be written as [5, 13]: \[\begin{aligned} \Delta\tilde{A}_r =& \frac{1}{2}\,t M^2 \mathcal{T}\mathcal{D} +\frac{u_0}{2!}\,M^4 \mathcal{D}^2 \mathcal{U} +\frac{a_{05}}{5!}\,M^5 \mathcal{D}^{5/2}\mathcal{V}\mathcal{U} +\frac{a_{06}}{6!}\,M^6 \mathcal{D}^3 \mathcal{U}^{3/2}\notag\\ &+ \frac{a_{14}}{4!}\,t M^4 \mathcal{T}\mathcal{D}^2 \mathcal{U}^{1/2} +\frac{a_{22}}{2!2!}\,t^2 M^2 \mathcal{T}^2 \mathcal{D}\mathcal{U}^{-1/2} -\frac{1}{2}\,t^2 \mathcal{K}, \label{eq:5} \end{aligned} \tag{4}\] where \(t\) and \(M\) are temperature-like and density-like variables respectively related to \(\Delta\tilde{T}\) and \(\Delta\tilde{\rho}\) defined by:
\[t = c_t \Delta\tilde{T} + c\left(\frac{\partial \Delta\tilde{A}}{\partial t}\right)_M \label{eq:6} \tag{5}\] \[M = c_{\rho}\left(\Delta\tilde{\rho} – d_1 \Delta\tilde{\rho}\right) + c\left(\frac{\partial \Delta\tilde{A}}{\partial M}\right)_t \label{eq:7} \tag{6}\] with the corresponding transformation: \[\Delta\tilde{A} = \Delta\tilde{A}_r – c\left(\frac{\partial \Delta\tilde{A}}{\partial t}\right)_M \left(\frac{\partial \Delta\tilde{A}}{\partial M}\right)_t \label{eq:8} \tag{7}\] where, \(c_t\), \(c_{\rho}\) and \(d_1\) are system-dependent constants. The coefficient \(c\) arises because of the vapor-liquid asymmetry of fluids. Thus, the coefficient \(c\) is also system-dependent parameter that mixes the field variables \(t\) and \(M\). The coefficients \(a_{05}\), \(a_{06}\), \(a_{14}\), \(a_{22}\) are system-dependent parameters and \(u_0 = u \Lambda\), where \(\Lambda\) is a dimensionless cutoff wave number and \(u\) is defined below, whilst the crossover functions \(\mathcal{T}\), \(\mathcal{D}\), \(\mathcal{U}\), \(\mathcal{V}\) and \(\mathcal{K}\) are defined by:
\[\mathcal{T} = \mathcal{Y}^{(2-1/\nu)/\omega}, \qquad \mathcal{D} = \mathcal{Y}^{-\eta/\omega}, \qquad \mathcal{U} = \mathcal{Y}^{1/\omega}, \label{eq:9a} \tag{8}\]
\[\mathcal{V} = \mathcal{Y}^{(2\omega_a-1)/\omega}, \qquad \mathcal{K} = \frac{\nu}{\alpha \bar{u}\Lambda} \left[\mathcal{Y}^{-\alpha/(\nu\omega)} – 1\right]. \label{eq:9b} \tag{9}\]
In terms of a crossover function, \(\mathcal{Y}\) to be determined from \[1 – (1 – \bar{u})\mathcal{Y} = u\left(1 + \frac{\Lambda^2}{\kappa^2}\right)^{1/2} \mathcal{Y}^{1/\omega}, \label{eq:10} \tag{10}\] with \[\kappa^2 = t\mathcal{T} + \frac{1}{2}\,u\Lambda M^2 \mathcal{D}\mathcal{U}, \label{eq:11} \tag{11}\] with \(\bar{u} = u/u^*\). In these expressions \(\nu\), \(\eta\), \(\omega\) and \(\omega_a\) are universal critical exponents, \(u^*\) is another universal constant, called the fixed-point coupling constant found elsewhere [5, 13]. The values of the critical exponents that will be used in this work are presented in Table 1.
The mixing of the field variables \(t\) and \(M\) results in an asymmetry as well as in a presence of a singularity the coexistence curve diameter of fluids. The deduction of the density is straightforwardly in verted from Eq. (6). Therefore, the densities on the liquid and vapor can be deduced from the present work as [12]: \[\rho_{\text{liq.,vap.}} = \rho_c \left[ 1 + \frac{1}{c_{\rho}} \left\{ M – c\left(\frac{\partial \Delta\tilde{A}_r}{\partial t}\right)_M \right\} + d_1 \Delta\tilde{T} \right]_{\text{liq.,vap.}}. \label{eq:12} \tag{12}\]
Near the critical point and along the along the coexistence curve \(\rho=\rho_{\text{cxc}}\), the renormalization-group theory predicts that [12, 13]: \[\frac{\rho_{\text{liq}}+\rho_{\text{vap}}}{2\rho_c} = 1 + d_1 \Delta\tilde{T} + d_{s1}\Delta\tilde{T}^{(1-\alpha)} + d_{s2}\Delta\tilde{T}^{(1-\alpha+\Delta)} +\cdots, \label{eq:13} \tag{13}\] where \(\rho_c\), is the critical density and \(\Delta\tilde{T}\) is the reduced temperature. The constant \(d_1\), \(d_{s1}\) and \(d_{s2}\) are the linear term, the \((1-\alpha)\), and the \((1-\alpha+\Delta)\) terms respectively, while the constant \(\alpha\) is the critical exponent that characterizes the divergence of the specific heat at constant volume, \(\beta\), \(\Delta\), are other critical exponents given in Table 1.
| Constant | Value |
|---|---|
| \(\alpha\) | 0.11 |
| \(\eta\) | 0.033 |
| \(\nu\) | 0.63 |
| \(\Delta\) | 0.51 |
| \(\omega=\Delta/\nu\) | 0.80952 |
| \(\omega_a\) | 2.1 |
| \(u^*\) | 0.472 |
Since coefficient \((1-\alpha)\) is close to unity, which makes the singularity in the coexistence-curve diameter Eq. (12) very hard to spot. Whereas, previous experiments carried-out by Weiner, Langley and Ford on sulfurhexafluoride [15] whose data were later assessed by Ley-Koo and Green [16], suggest the existence of the singular term in Eq. (12). Afterward, other experiments carried-out by Pestak et al. [17] on nitrogen, neon, and on ethane and ethylene [17] have also validated the presence of the hook in the rectilinear diameter identified by the weak singular term corresponding to the exponent \((1-\alpha)\). The crossover model reproduces this expansion with the coefficients \(d_{s1}\) and \(d_{s2}\) given by the following expressions, with a new exponent \(\Delta=\omega\nu\):
\[d_{s1} = -9.547\, c_t^{(1-\alpha)} (\Lambda\bar{u})^{-3(2\nu-1)} \frac{c}{c_{\rho}}, \label{eq:14} \tag{14}\] \[d_{s1} = 8.820\, c_t^{(1-\alpha+\Delta)} (1-\bar{u}) (\Lambda\bar{u})^{-3(2\nu-1)-2\Delta} \frac{c}{c_{\rho}}. \label{eq:15} \tag{15}\]
The coexisting liquid and vapor densities are represented by the relation: \[\frac{\rho_{\text{liq}}-\rho_{\text{vap}}}{2\rho_c} = A_{\beta}\left|\Delta\tilde{T}\right|^{\beta} +\cdots. \label{eq:16} \tag{16}\]
The value of critical exponent \(\beta\) is listed on Table 1. The coefficient \(A_{\beta}\) can be determined from the model or by fitting the experimental data to Eq. (16). The expression of the most dominant coefficient in Eq. (16) as calculated from the crossover model and is presented as follow: \[A_{\beta} = \left[ \frac{2(1+x)}{x^{2\beta}} \left( \frac{(\Lambda\bar{u})^{-2(2-1/\nu)\beta-\eta}}{u^*} \right) \right]^{1/2} \frac{c_t^{\beta}}{c_{\rho}}, \label{eq:17} \tag{17}\] with \[x = \frac{ 2(3\nu-1) – \sqrt{ 4-54\nu+9(9-u^*)\nu^2 } }{ 10-3\nu(5-u^*) }. \label{eq:18} \tag{18}\]
Although n-heptane is relatively inadequate as a fuel, it is largely used as a solvent. It is a major component of rubber cement and is sold commercially for use in paints and for fast-drying lacquers and glues. In this latter application, n-heptane acts to dissolve pigments and make its coloration uniformly. It is also used in pneumatic paint sprayers. In calibration applications, n-heptane is used as a reference fluid for saturated liquid heat capacities and liquid densities around \(700~\mathrm{kg\,m^{-3}}\) [18] because of the industrial importance of n-heptane; this work sheds more light on the importance of its thermodynamic properties.
For a comparison of our crossover model with experimental data of n-heptane, we consider the coexistence-curve diameter of n-heptane and the saturated liquid and vapor densities of n-heptane based on an equation of state in the critical region previously formulated by Ladjama [19]. The system-dependent constants in the crossover model [1, 3] were determined from a fit to the \(P\)–\(\rho\)–\(T\) data of Kurumov [20] with temperature and density ranges of \(550~\mathrm{K} \le T \le 530~\mathrm{K}\), and \(128~\mathrm{kg\,m^{-3}} \le \rho \le 270~\mathrm{kg\,m^{-3}}\) respectively. The critical parameters adopted in this work for n-heptane are those measured by Kurumov [20] and adopted by Ladjama [19]; they are as follows:
\[T_c = 540.13~\mathrm{K}, \quad P_c = 2.376~\mathrm{MPa}, \quad \rho_c = 232~\mathrm{kg/m^3}, \label{eq:19} \tag{19}\]
The fixed system-dependent parameters [19] used to compute the liquid and vapor densities of n-heptane are presented in Table 2. A comparison of the the saturated vapor and liquid densities computed from the crossover model equation of state [19] with those generated by the REFPROP package [21] with the experimental rectilinear diameter data of n-heptane as a function of reduced temperature reported by Abdulagatov et al. [22] with the saturated temperatures and the saturated densities on the coexistence curve derived from the isochoric heat capacity measurements of n-heptane and those measured by Artyukhovskaya et al. [23] is shown in Figure 1. In addition, we show the reduced densities of the coexisting vapor and liquid phases of n-heptane as a function of reduced temperature reported by Abdulagatov et al. [22] and Artyukhovskaya et al. [23] in Figure 2 and Figure 3, with more details are presented in Figure 4 in the region very near the critical point. The data reported by Abdulagatov et al. [22] and Artyukhovskaya et al. [23] are in a quite satisfactory agreement when compared to the crossover model and with those data generated from REFPROP [21].
It is worth to notice that the offset of the Artyukhovskaya et al. [23] data at higher temperature with respect to the curve predicted from the crossover model equation is probably due to the fact that these density-data were obtained from optical and microfloat measurements of the density in thin layers of matter near the critical point. The agreement of the Abdulagatov et al. [22] and Artyukhovskaya et al. [23] sets of data with the predicted coexistence curves are typically measured on the level of a few tenths % uncertainties (No experimental uncertainties were reported for the data measured by Abdulagatov et al. [22], while those measured by Artyukhovskaya et al. [23] the experimental uncertainties for temperatures and densities are quoted to be \(\sigma_T = 0.001\), \(\sigma_{\rho} = 5 \cdot 10^{4} ~\mathrm{g/cm^3}\)) and because Figure 5 is too roughly scaled to make any conclusions about the goodness of the comparison, especially very close to the critical point, it is therefore, more adequate and even more revealing to show deviation plots. Figure 5 shows the deviation plots of Abdulagatov et al. [22] and Artyukhovskaya et al. [23] experimental data with respect to the values calculated from the crossover model of Eq. (16).
| Critical parameters | \(T_c = 540.13~\mathrm{K},\; P_c = 2.736~\mathrm{MPa},\; \rho_c = 232.00~\mathrm{kg/m^3}\) |
| Crossover parameters | \(\bar{u} = 0.464,\; \Lambda = 1.734\) |
| Scaling-field parameters | \(c_t = 2.541,\; c_\rho = 2.259,\; c = -0.009\) |
| Pressure background parameters | \(\tilde{A}_0 = -1,\; \tilde{A}_1 = -7.15,\; \tilde{A}_2 = 10.12,\; \tilde{A}_3 = 5.582,\; \tilde{A}_4 = -1.340,\; d_1 = -0.964\) |
| Classical parameters | \(a_05 = -0.035,\; a_06 = 0.313,\; a_14 = 1.562,\; a_22 = 1.481\) |
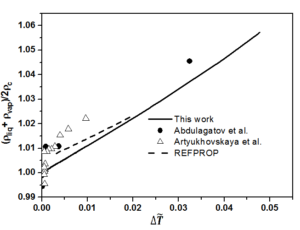
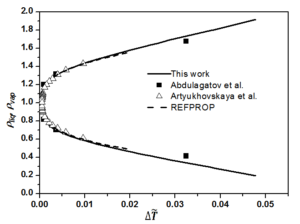
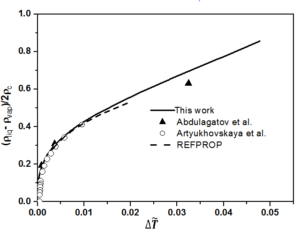
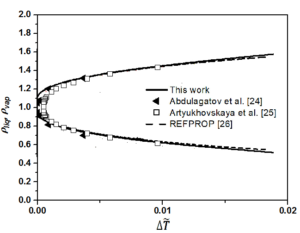
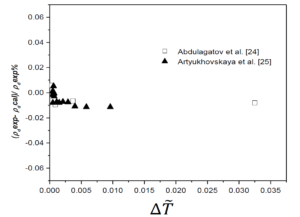
The present work compares the computed liquid and vapor densities of normal heptane from the EOS crossover model, without any fitting to the liquid-vapor phase experimental data reported by Abdulagatov et al. [22] and those measured by Artyukhovskaya et al. [23] or the with the generated saturated densities from the REFPROP package [21]. In addition, it is worth noticing that the crossover model implies a value of the specific-heat-amplitude ratio \(A^{+}/A^{-}\), which is about 5% below the theoretical value, that is the best ratio as obtained from renormalization-group calculations for the Ising model [7, 8], and because the amplitude of the hook of the coexistence-curve diameter is firmly related to the specific-heat amplitude \(A^{-}\). Therefore, it is sometimes very difficult to make any statement about the magnitude of this hook as long as the amplitude ratio is not determined with great certainty.
The authors wish to thank Eric Lemmon for sending their results for normal heptane and the link to REFPROP package .
This work was supported by the Direction Générale de la Recherche Scientifique et Technologique (DGRST) of the Algerian government under grand MESRS/FNR/2011.
Fisher, M. E. (1982). In Critical Phenomena (F. J. W. Hahne, Ed., Lecture Notes in Physics, Vol. 186, pp. 1–139). Springer.
Greer, S. C., & Moldover, M. R. (1981). Thermodynamic anomalies at critical points of fluids. Annual Review of Physical Chemistry, 32(1), 233-265.
Sengers, J. L., Morrison, G., & Chang, R. F. (1983). Critical behavior in fluids and fluid mixtures. Fluid Phase Equilibria, 14, 19-44.
Kumar, A., Krishnamurthy, H. R., & Gopal, E. S. R. (1983). Equilibrium critical phenomena in binary liquid mixtures. Physics Reports, 98(2), 57-143.
Abbaci, A. (2005). Thermodynamic properties of ethane in the critical region. Journal of Molecular Liquids, 118(1-3), 31-36.
Abbaci, A., & Berrezeg, A. (2004). A thermodynamic equation of state for the critical region of ethylene. International Journal of Thermophysics, 25(3), 739-752.
Wilson, K. G., & Kogut, J. (1974). The renormalization group and the \(\epsilon\) expansion. Physics Reports, 12(2), 75-199.
Nicoll, J. F., & Albright, P. C. (1985). Crossover functions by renormalization-group matching: three-loop results. Physical Review B, 31(7), 4576.
Nicoll, J. F., Chang, T. S., & Stanley, H. E. (1974). Nonlinear solutions of renormalization-group equations. Physical Review Letters, 32(25), 1446.
Bagnuls, C., & Bervillier, C. (1985). Nonasymptotic critical behavior from field theory at \(d=3\): The disordered-phase case. Physical Review B, 32(11), 7209.
Patashinskii, A. Z., & Potrovskii, V. I. (1979). Fluctuation Theory of Phase Transitions. Pergamon Press.
Ladjama, S., & Abbaci, A. (2017). Phase diagram of argon: The prediction of the coexisting liquid and vapor densities of argon near the critical point. The European Physical Journal Special Topics, 226(5), 967-975.
Abbaci, A. (2004). Liquid-Vapor Density of Sulfur Hexafluoride in the Critical Point. Iranian Journal of Chemistry and Chemical Engineering, 23(1), 103-108.
Abbaci, A., Berrezeg, A., & Samar, M. E. H. (2003). Prediction of the coexisting liquid and vapour densities of ethylene near the critical point. High Temperatures-High Pressures, 35(36), 691-697.
Weiner, J., Langley, K. H., & Ford Jr, N. C. (1974). Experimental evidence for a departure from the law of the rectilinear diameter. Physical Review Letters, 32(16), 879.
Ley-Koo, M., & Green, M. S. (1977). Revised and extended scaling for coexisting densities of S F 6. Physical Review A, 16(6), 2483.
Pestak, M. W., Goldstein, R. E., Chan, M. H. W., de Bruyn, J. R., Balzarini, D. A., & Ashcroft, N. W. (1987). Three-body interactions, scaling variables, and singular diameters in the coexistence curves of fluids. Physical Review B, 36(1), 599.
Span, R., & Wagner, W. (2003). Equations of state for technical applications. I. Simultaneously optimized functional forms for nonpolar and polar fluids. International Journal of Thermophysics, 24(1), 1-39.
Ladjama, S. (2018). Title of the doctoral dissertation (Doctoral dissertation, Université Badji Mokhtar, Annaba).
Kurumov, D. S. (1990). Thermodynamic parameters of hydrocarbons in the critical range: 1. n-octane and n-nonane. Journal of Engineering Physics, 59(6), 1528-1532.
National Institute of Standards and Technology. (2023). Nist Chemistry Webbook (NIST Standard Reference Database No. 69).
Abdulagatov, I. M., Rabinovich, V. A., & Dvoryanchikov, V. I. (1999). Thermodynamic Properties of Fluids and Fluid Mixtures. Begell House.
Artyukhovskaya, L. M., Shimanskaya, E. T., & Shimanskii, V. I. (1973). The coexistence curve of heptane near the critical point. Soviet Physics—JETP, 36, 1140-1142.